

Fields of Research: PHIP
 |
 |
Ortho- and Parahydrogen:
Spin Isomers of Molecular Hydrogen
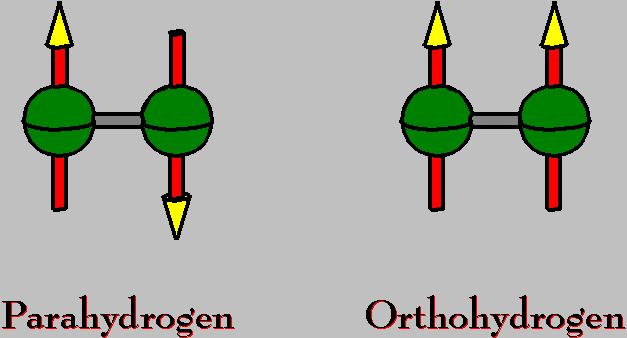
| 1H atom | I = 1/2 | I = nuclear spin |
| H2 molecule | I = 0 | Parahydrogen (singlet state) |
| I = 1 | Orthohydrogen (triplet state) |
Molecular hydrogen occurs in two isomeric forms, namely with its two proton spins aligned either parallel (orthohydrogen) or antiparallel (parahydrogen). In the state of thermal equilibrium at room temperature dihydrogen contains 25 % of parahydrogen (nuclear singlet state) and 75 % of orthoghydrogen (nuclear triplet state). This behavior reflects the threefold degeneracy of the triplet state, and the almost equal population of the energy levels at room temperature as demanded by the Maxwell-Boltzmann distribution.
Ortho-/Parahydrogen enrichment
The PHIP phenomenon can be observed during hydrogenation by using either enriched ortho- or parahydrogen. Using orthohydrogen the sequence of emission and absorption is reversed relative to using enriched parahydrogen. It is possible to enrich both spin isomers, whereby the degree of enrichment can be determined accurately via thermal conductivity cells.
The mixture can be enriched in parahydrogen by making use of the fact that, in the presence of a paramagnetic adsorber (active charcoal), a thermal equilibrium is established between ortho- and parahydrogen. At low temperatures, therefore, parahydrogen is favored energetically. In this way our apparatus produces a constant flow of a mixture of 51 % parahydrogen and 49 % orthohydrogen at 77 K (liquid nitrogen).
The mixture can be enriched in ortho hydrogen by expoiting the fact that, at low temperatures, the two spin isomers are not equally adsorbed on diamagnetic surfaces (theory of the hindered rotator). We use single stage separation of orthohydrogen on g-alumina at 77 K, and obtain a mixture of 15 % parahydrogen and 85 % orthohydrogen.
Last modified Monday, 16.9.2002 +++ webdesign & webmaster: klaus blömeke